The Levi Laboratory is proud to announce a publication in The Journal of Immunology entitled “Tuning Macrophage Phenotype to Mitigate Skeletal Muscle Fibrosis.” Based on clinical observations of post-traumatic muscular fibrosis, we developed and validated a novel mouse model of muscular polytrauma via ischemia/reperfusion and cardiotoxin antecedent to aberrant, fibrotic healing. This work delineates the role of macrophage derived TGF-beta through single cell RNA sequencing and large confocal microscopy scans facilitated by the collaboration and infrastructure of the University of Michigan research community. Our group continues to be grateful for the partnership between expert investigators from Stanford University (Dr. Michael Longaker), Acceleron Pharmaceuticals (Dr. Ravi Kumar), and intrauniversity departments like the Department of Computational Medicine and Bioinformatics (Dr. Jun Li and Dr. Simone Marini).
This work further highlights several young investigators including Dr. David Stepien, Charles Hwang, Dr. Simone Marini, and Chase Pagani. This work will inform our future studies on post-injury muscular fibrosis and provide new targets and insights for our investigation of this disease. Special thank yous to the University of Michigan Department of Surgery and Section of Plastic Surgery, Center for Molecular Imaging, and the rest of the BWR laboratory team.
PMID: 32161098. (Read here!)
Abstract
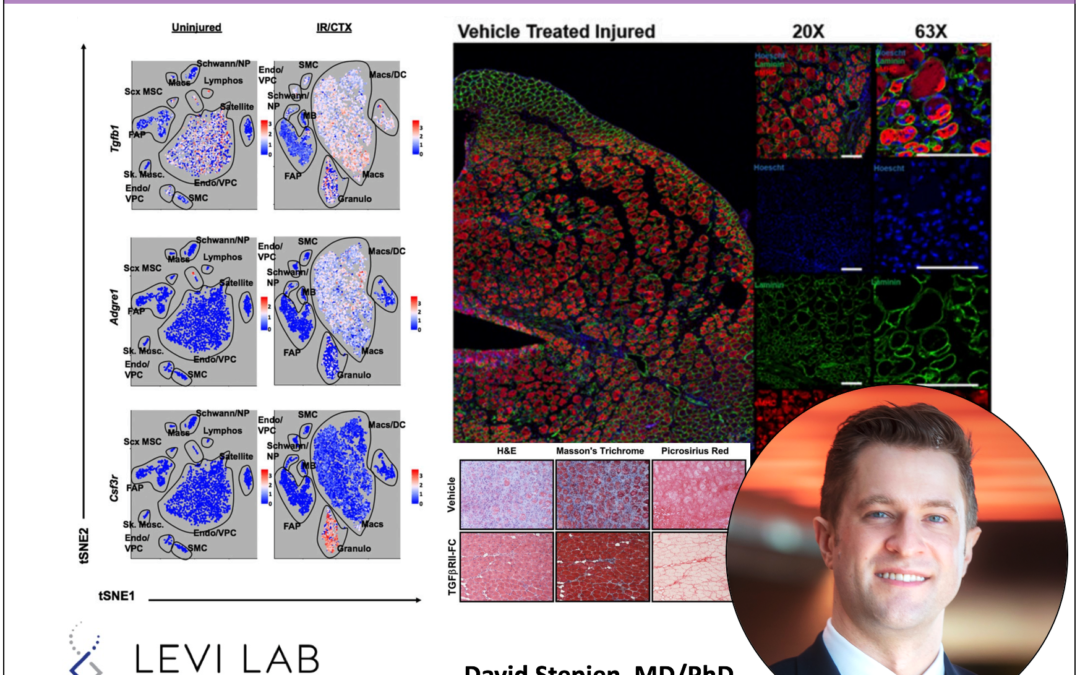
Myeloid cells are critical to the development of fibrosis following muscle injury; however, the mechanism of their role in fibrosis formation remains unclear. In this study, we demonstrate that myeloid cell-derived TGF-β1 signaling is increased in a profibrotic ischemia reperfusion and cardiotoxin muscle injury model. We found that myeloid-specific deletion of Tgfb1 abrogates the fibrotic response in this injury model and reduces fibro/adipogenic progenitor cell proliferation while simultaneously enhancing muscle regeneration, which is abrogated by adaptive transfer of normal macrophages. Similarly, a murine TGFBRII-Fc ligand trap administered after injury significantly reduced muscle fibrosis and improved muscle regeneration. This study ultimately demonstrates that infiltrating myeloid cell TGF-β1 is responsible for the development of traumatic muscle fibrosis, and its blockade offers a promising therapeutic target for preventing muscle fibrosis after ischemic injury.